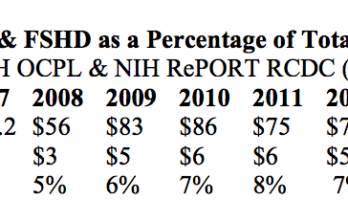The San Diego-based biotech company, aTyr Pharma, Inc., released findings on March 30, 2016, from a Phase 1b/2 clinical trial of its experimental therapy Resolaris in adult facioscapulohumeral muscular dystrophy (FSHD) patients. While… Read More »
aTyr Pharma to advance Resolaris™ clinical programs
Good times! TD Garden Tournament
Our first-ever basketball tournament, at Boston’s legendary TD Garden–home of the Boston Celtics–was a rousing success. Teams from Acceleron Pharma, SHIFT Communications, O’Connor Drew, Mouse Specifics, Social Sports Boston and… Read More »
FSH Society submits testimony to U.S. Congress
FSH Society President & CEO Daniel Perez has submitted his annual testimony to the U.S. House Appropriations Committee, Subcommittee on Labor, Health and Human Services, Education and Related Agencies (LHHSE)…. Read More »
Publication highlights release of muscular dystrophy action plan
The Muscular Dystrophy Coordinating Committee (MDCC), a congressionally authorized group of representatives from federal agencies and patient advocates, recently released an updated version of its Action Plan for the Muscular… Read More »
NIH funding for Facioscapulohumeral muscular dystrophy sets new record
We have some good news to share on National Institutes of Health funding for FSHD. As part of the FSH Society’s committee work in Washington, DC, we recently received FY2015… Read More »