Donor Impact
Your support has propelled us forward.
Four years ago, fueled by your generosity and commitment, we launched a bold plan to accelerate therapeutic development for FSHD while empowering a growing, active community. We laid out a roadmap with lofty goals and aggressive milestones.
Together we have reached every mile marker ahead of schedule.
Advancing Research
Our ability to advance research grew, through continued investment in specific research projects, advancing Therapeutic Accelerator initiatives, and by bringing together an ever-growing network of stakeholders to collaborate, problem solve, and chart the best course forward.
- Direct research funding was utilized to validate blood biomarkers and use AI to identify risk factors for symptom progression.
- The TestFSHD initiative advanced trial-readiness for our community by providing free genetic testing for 150 patients.
- The True Cost of FSHD study gathered information from over 300 households about FSHD-related expenses, so that we can better advocate for insurance coverage of future treatments.
- Data is currently being analyzed; results are expected to be published in 2023.
- The 29th annual International Research Congress on FSHD brought together researchers and clinicians from all over the world for collaboration and inspiration.
- The Industry Collaborative Workshop congregated key stakeholders to review progress in therapy development and agree on the best course forward.


Expanding & Deepening Community
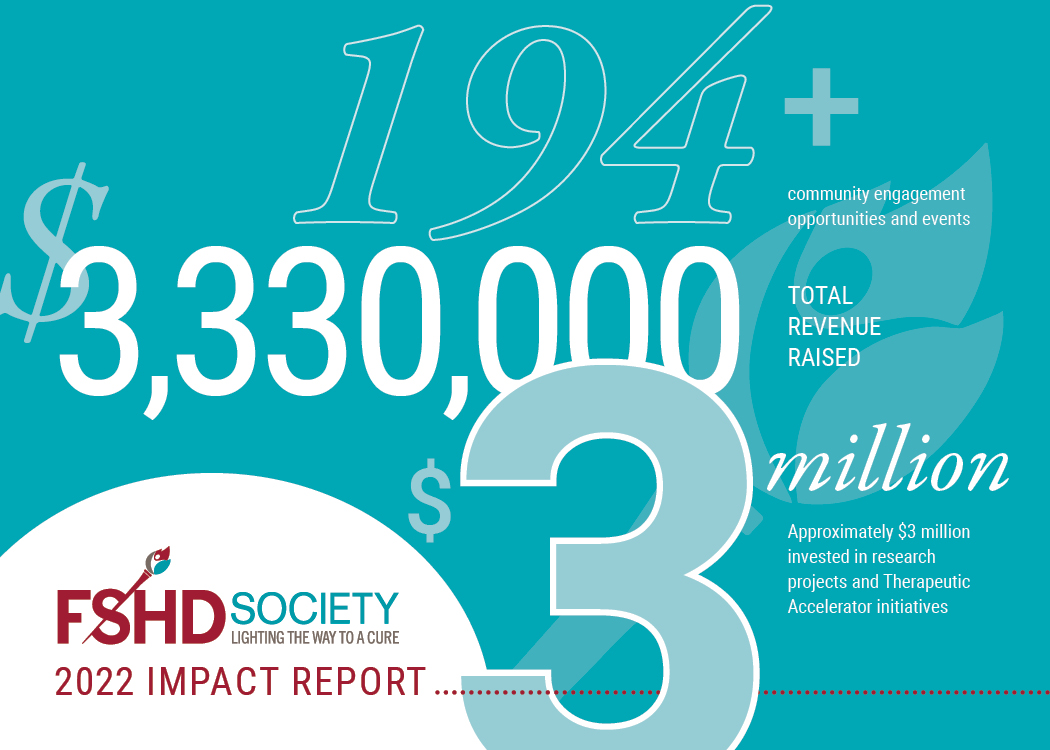
194+ opportunities to engage with your community and build meaningful connections through a variety of channels, including:
- Gathering Place virtual affinity groups, meeting monthly
- Twice monthly FSHD Radio podcasts
- Local Chapter meetings (including in-person, virtual, and hybrid gatherings), connecting people in their home region
- Monthly educational webinars through FSHD University
- The bi-annual FSHD Patient Connect Conference and quarterly regional FSHD 360 conferences
25% of mission funding was raised through volunteer-led events and local efforts
Our worldwide community grew by 15%, including nearly 600 patients, who’ve self-identified as being ready & willing to participate in research

