To crack the code of FSHD, patients are absolutely essential
All of the breakthroughs—the discovery of the genetic causes, understanding why some patients vary so greatly in the severity of their symptoms, teasing out the biochemical pathways that could point to future treatments—were made because patients stepped up to the plate.
Too often, we hear patients say they’ll volunteer when there’s a treatment. But we will never get to a treatment unless patients participate in fundamental research now. FSHD is uniquely human, so no laboratory mouse can ever fully model the disease. The genetic “package” that causes FSHD is found only in people. We owe an enormous debt to the patients who give DNA samples. Who submit to long interviews and exhausting physical tests. Allow a surgeon to cut out a small muscle sample. Who fight claustrophobia to lie in the narrow bore of an MRI machine.
Equally important are patients’ family members, both affected and unaffected, who provide the best experimental controls because of their shared genetic and environmental backgrounds. A parent or sibling who has very mild symptoms may hold the key to understanding the factors that protect against the full-blown development of FSHD symptoms in a more severely affected family member.
We are more hopeful today than ever before that a treatment is within sight. We cannot guarantee when that treatment will arrive, but here’s one thing we guarantee: If you volunteer for research, your participation will without question help move us a step closer to that day.
Scientific Overview of FSHD
Read the latest on wikipedia
Glossary of Scientific Terms
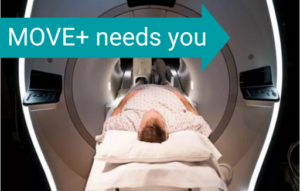
Is MRI the “holy grail” for FSHD clinical trials?
The MOVE+ study aims to find out. Participants needed! by June Kinoshita, FSHD Society Imagine that you are in an FSHD clinical trial a few years in the future. On… Read More »
Vita Therapeutics’ cell-based therapy update
The company switches up its strategy and plans to develop muscle stem cells that evade the immune system by June Kinoshita, FSHD Society Hot off the press is this news… Read More »
From the Wayback Machine, 2010
Who hasn’t worried that when they clean out the attic, they’ll inadvertently send a precious heirloom to the trash heap? Sometimes a good Samaritan finds the treasure and returns it…. Read More »
FSHD Society partnering in €21 Million project
Aiming to accelerate disease treatment and care for rare diseases See PaLaDIn Launch Press Release The FSHD Society is embarking on an exciting international collaboration to speed up the development… Read More »