What does this mean for patients?
An FDA Fast Track designation DOES NOT mean that the drug has been approved for use by patients. AOC 1020 is still a long way from that, but the FDA has approved a faster path through the new drug application and clinical trial process. It’s like getting TSA Pre to go through the security line at the airport more quickly. To find out more about what the trial involves, where the trial will be conducted, and the process for enrolling, be sure to sign up for our webinar about the FORTITUDE trial on February 14, 2023
Avidity Biosciences Granted FDA Fast Track Designation for AOC 1020 for the Treatment of Facioscapulohumeral Muscular Dystrophy
SAN DIEGO, Jan. 18, 2023 /PRNewswire/ — Avidity Biosciences, Inc. (Nasdaq: RNA), a biopharmaceutical company committed to delivering a new class of RNA therapeutics called Antibody Oligonucleotide Conjugates (AOCs™), today announced that the U.S. Food and Drug Administration (FDA) has granted Fast Track designation to AOC 1020 for the treatment of facioscapulohumeral muscular dystrophy (FSHD). FSHD is a serious, rare, hereditary muscle-weakening condition marked by life-long, progressive loss of muscle function that causes significant pain, fatigue, and disability. AOC 1020 is being studied in the Phase 1/2 FORTITUDE™ clinical trial in adults with FSHD and is the company’s second muscle-targeting small interfering RNA (siRNA) AOC in clinical development. Avidity plans to share data from a preliminary assessment of AOC 1020 in approximately half of study participants from the FORTITUDE trial in the first half of 2024.
Fast Track designation enables more frequent interactions with the FDA to expedite the development and review process for drugs intended to treat serious or life-threatening conditions and that demonstrate the potential to address unmet medical needs. Currently, there are no FDA-approved treatments for people living with FSHD.
“The FDA Fast Track designation for AOC 1020 reinforces the importance of finding an effective treatment to help people living with FSHD, a devastating and debilitating muscular dystrophy disorder with no treatment options,” said Steve Hughes, M.D., chief medical officer at Avidity. “AOC 1020 is designed to directly target the disease-causing gene, DUX4, to address the underlying cause of FSHD. We look forward to working collaboratively with the FDA to bring the first RNA therapy directly targeting DUX4 to patients as quickly as possible.”
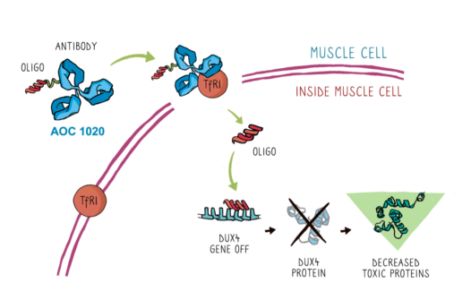 Avidity’s proprietary AOCs are designed to combine the specificity of monoclonal antibodies (mAbs) with the precision of oligonucleotide therapies to target the root cause of diseases previously untreatable with RNA therapeutics. AOC 1020 consists of a proprietary mAb that binds to the transferrin receptor 1 (TfR1) conjugated with a siRNA targeting double homeobox 4 (DUX4) mRNA. The abnormal expression of DUX4 protein leads to changes in gene expression in muscle cells that are associated with the life-long, progressive loss of muscle function in patients with FSHD.
Avidity’s proprietary AOCs are designed to combine the specificity of monoclonal antibodies (mAbs) with the precision of oligonucleotide therapies to target the root cause of diseases previously untreatable with RNA therapeutics. AOC 1020 consists of a proprietary mAb that binds to the transferrin receptor 1 (TfR1) conjugated with a siRNA targeting double homeobox 4 (DUX4) mRNA. The abnormal expression of DUX4 protein leads to changes in gene expression in muscle cells that are associated with the life-long, progressive loss of muscle function in patients with FSHD.
Avidity has three distinct rare disease programs in the clinic. In addition to AOC 1020, the company is also evaluating AOC 1001 in the Phase 1/2 MARINA™ and MARINA open-label extension (MARINA-OLE™) clinical trials for the treatment of myotonic dystrophy type 1 (DM1) and AOC 1044 in the Phase 1/2 EXPLORE44™ trial for the treatment of Duchenne muscular dystrophy (DMD) mutations amenable to exon 44 skipping (DMD44).
The FORTITUDE™ Phase 1/2 Trial of AOC 1020 in Adults with FSHD
The FORTITUDE™ trial is a randomized, placebo-controlled, double-blind, Phase 1/2 clinical trial designed to evaluate AOC 1020 in approximately 70 adult participants with FSHD. FORTITUDE will evaluate the safety, tolerability, pharmacokinetics, and pharmacodynamics of AOC 1020 administered intravenously, with the primary objective being the safety and tolerability of AOC 1020 in FSHD patients. Activity of AOC 1020 will be assessed using key biomarkers, including magnetic resonance imaging (MRI) measures of muscle volume and composition. Though the Phase 1/2 trial is not statistically powered to assess functional benefit, it will explore the clinical activity of AOC 1020 including measures of mobility and muscle strength as well as patient reported outcomes and quality of life measures. Participants will have the option to enroll in an open-label extension study at the end of the treatment period in the FORTITUDE study.
About AOC 1020
AOC 1020 is designed to treat the underlying cause of FSHD, which is caused by the abnormal expression of a gene called double homeobox 4 or DUX4. The abnormal expression of DUX4 protein leads to changes in gene expression in muscle cells that are associated with the life-long, progressive loss of muscle function in patients with FSHD. AOC 1020 aims to reduce the expression of DUX4 mRNA and DUX4 protein in muscles in patients with FSHD. AOC 1020 consists of a proprietary monoclonal antibody that binds to the transferrin receptor 1 (TfR1) conjugated with a siRNA targeting DUX4 mRNA. In preclinical studies, a single intravenous dose with the murine version of AOC 1020 prevented development of muscle weakness demonstrated by three functional assays – treadmill running, in vivo force and compound muscle action potential. AOC 1020 is currently in Phase 1/2 development as part of the FORTITUDE™ trial in adults with FSHD.
Read the full press release here


Hello!
The twins Fazit and Nazip, born in 1968, Razit, born in 1972, are writing to you. We have been diagnosed with FSHD1. We would be happy to participate in clinical trials of new drugs for the treatment of FSHD1 You are our last hope, we will be very grateful to you.
Thank you in advance!
Fazit, Nazip, Razit